Why Menopause Disrupts Vaginal Bacterial Balance
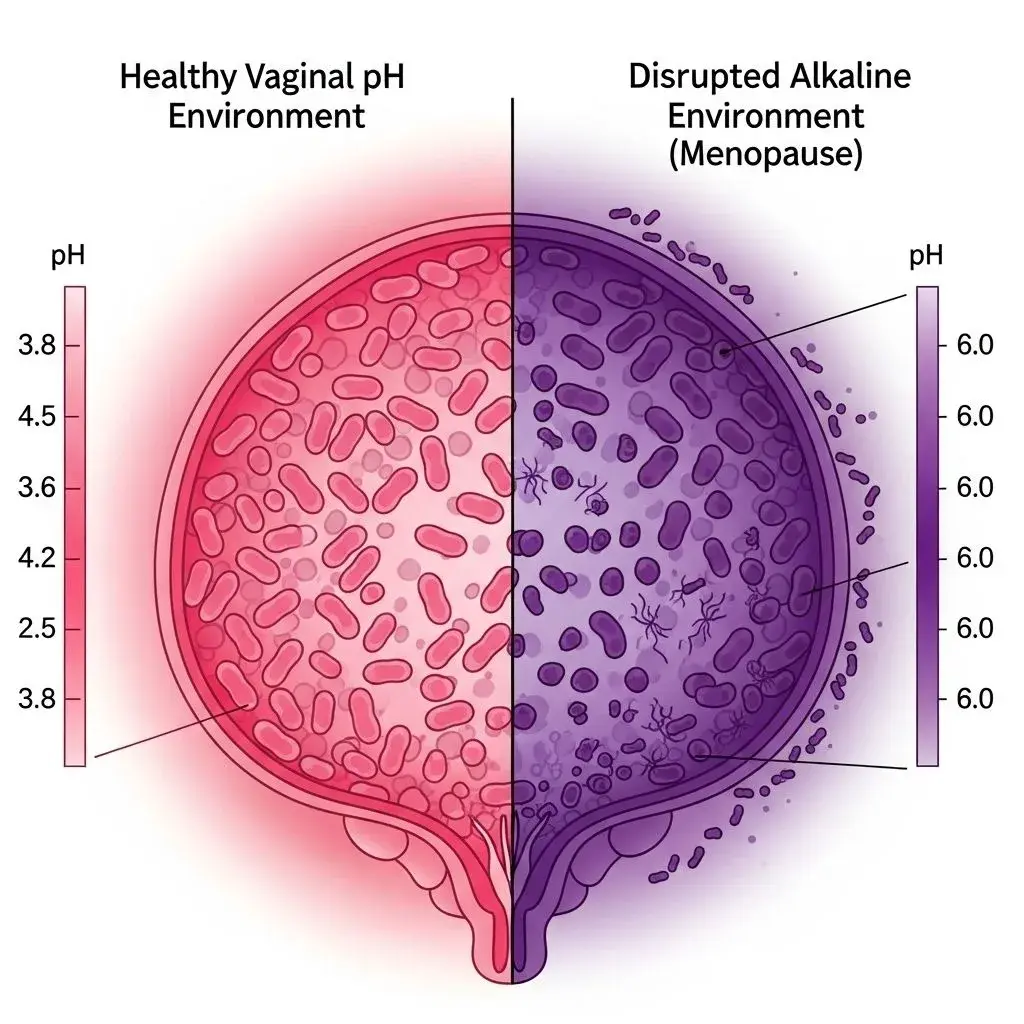
The question can perimenopause cause BV has a clear answer rooted in estrogen's crucial but underappreciated role in vaginal health. Estrogen maintains glycogen stores in vaginal epithelial cells, which lactobacilli bacteria metabolize into lactic acid, creating the acidic pH (3.8-4.5) that prevents pathogenic bacteria overgrowth. When estrogen declines during perimenopause and menopause, glycogen production drops by 40-60%, lactobacilli populations plummet, and vaginal pH rises to 5.5-7.0. This creates ideal conditions for bacterial vaginosis menopause cases to surge, with studies showing BV prevalence increases from 15% in premenopausal women to nearly 30% in postmenopausal women.
What makes this particularly challenging is the cascade effect: elevated pH not only allows BV-associated bacteria like Gardnerella vaginalis to thrive, but also creates vulnerability to recurrent infections. Each BV episode further depletes beneficial lactobacilli, making subsequent infections more likely and harder to resolve. Meanwhile, the same estrogen decline causes vaginal tissue thinning (atrophy), reducing physical barrier protection and creating micro-abrasions that facilitate bacterial invasion. This explains why many women experience their first-ever BV case during perimenopause despite decades of stable vaginal health.
The answer to does menopause cause yeast infections involves a different mechanism but similar hormonal trigger. Yeast infections actually decrease in frequency after menopause because Candida thrives in estrogen-rich environments. However, perimenopause creates the worst of both worlds—fluctuating estrogen causes unpredictable pH swings and lactobacilli disruption while still providing enough estrogen to support occasional yeast overgrowth. Understanding how hormonal stress compounds these issues is explored in our guide to stress impacts during menopause.
Common Misconceptions About Vaginal Infections in Menopause
Myth 1: BV Means Poor Hygiene
The persistent stigma around bacterial vaginosis contradicts medical reality: BV is a pH-driven dysbiosis, not a hygiene issue or sexually transmitted infection. In fact, excessive washing and douching—attempts to be 'cleaner'—actually increase BV risk by further disrupting the already-compromised bacterial balance. Clinical studies show menopausal women with BV typically have better hygiene practices than those without, because they're overcorrecting for symptoms. The evidence is clear: BV during menopause is hormonal, not behavioral.
Myth 2: Probiotics Work the Same as Premenopausal Women
Here's what beginners misunderstand: oral probiotics containing lactobacilli can help restore vaginal flora in younger women, but effectiveness drops significantly after menopause. Without adequate estrogen to produce the glycogen that feeds lactobacilli, supplemented bacteria struggle to colonize and maintain populations. The trade-off most doctors don't explain: vaginal probiotic suppositories work better than oral forms in menopausal women, but require consistent use (often 2-3 times weekly indefinitely) rather than the one-course cure many expect.
Myth 3: Treating BV Once Solves the Problem
The contradiction between treatment expectations and reality: standard antibiotic therapy (metronidazole or clindamycin) clears BV in 80-90% of cases initially, but recurrence rates in menopausal women reach 50-70% within three months. Why? Antibiotics eliminate BV-causing bacteria but don't address the underlying estrogen deficiency that created the vulnerable environment. Without correcting the hormonal cause, you're treating symptoms repeatedly rather than resolving the root problem, which requires a different therapeutic approach entirely.
Evidence-Based Prevention and Treatment Strategies
The most effective prevention strategy for bacterial vaginosis during menopause addresses the hormonal cause directly: vaginal estrogen therapy. Low-dose vaginal estrogen (creams, tablets, or rings) restores glycogen production, lowers pH, and supports lactobacilli recolonization without systemic hormone exposure. Clinical trials show vaginal estrogen reduces BV recurrence by 60-70% compared to antibiotics alone. What experienced gynecologists emphasize: this is preventive treatment that works best when started before recurrent infections establish a cycle, not rescue therapy after multiple BV episodes.
Practical management combines multiple approaches:
- pH-balancing maintenance: Use pH-balanced intimate washes (pH 4.5-5.5) and avoid alkaline soaps, douches, and scented products. Consider boric acid suppositories (600mg) used 1-2 times weekly for pH maintenance between BV episodes.
- Targeted probiotic support: Choose vaginal suppositories containing Lactobacillus crispatus or L. rhamnosus (specific strains matter) rather than oral probiotics. Use after antibiotic treatment to support recolonization.
- Barrier protection during sex: Semen is alkaline (pH 7.2-8.0) and temporarily disrupts vaginal pH for hours after intercourse. Condom use or post-sex pH-balancing rinses reduce BV triggers, though this creates intimacy trade-offs many couples find difficult.
For recurrent cases resistant to standard treatment, extended antibiotic protocols (twice-weekly metronidazole gel for 4-6 months) combined with vaginal estrogen show better success rates than either alone. The critical insight: you're managing a chronic hormonal condition, not curing an acute infection, which requires adjusting expectations from permanent resolution to effective long-term control. Understanding how hormonal changes affect multiple body systems is detailed in our guide to skin care during hormonal changes.
When Standard BV Treatment Fails
Here's the edge case that derails conventional treatment: approximately 10-15% of menopausal women develop antibiotic-resistant BV or harbor biofilm-forming bacteria that standard metronidazole can't penetrate. These women experience immediate BV recurrence after every antibiotic course, sometimes within days. The solution requires biofilm-disrupting agents (boric acid or hydrogen peroxide vaginal gel) used before antibiotics, followed by extended suppressive therapy. However, this approach demands 6-12 months of continuous treatment, carries higher side effect risk (vaginal irritation, burning), and still fails in 20-30% of severe cases. The limitation rarely discussed: some women ultimately choose symptom management over cure-focused treatment when side effects outweigh benefits, using pH-balancing products and accepting mild chronic symptoms rather than pursuing aggressive protocols. This isn't treatment failure—it's informed preference that medical guidelines don't always accommodate, though quality of life often improves compared to endless treatment cycles.


